Vici has considerable experience developing a variety of orally administered formulations.
- Tablets, uncoated or coated
- Minitablets
- Capsules, powder-filled and coated multi-particulate
- Oral solutions/suspensions
- Oral granules or powder for reconstitution
- ODT (orally disintegrating tablets)
- Chewable tablets
Bioavailability and Pharmacokinetics
Understanding bioavailability is critical to developing safe and effective formulations. Vici has expertise in designing dosage forms that meet target physical and chemical specifications, and release the right amount of drug at the right location of the gastrointestinal tract. Vici employs a variety of formulation technologies to achieve a specific desired rate of drug release upon administration.
Increasing bioavailability of poorly soluble drugs, including BCS-2 and BCS-4 classified molecules. Techniques at Vici include:
- Micronization of poorly soluble drugs for improved bioavailability
- Creation of amorphous solid dispersion for improved solubility
- Improving bioavailability through the addition of solubility enhancing inactive ingredients.
- Once-a-day (QD) or twice-daily (BID) drug release formulations may be created for better patient convenience, compliance, and increased treatment efficacy by a variety of technologies.
- Rapid release of drug for fast onset of action.
- Enteric-coated formulations are used when the drug needs to bypass the stomach upon ingestion. This may be preferred for a variety of reasons including:
- Delaying onset of action of the drug
- Protecting the drug from the acidic gastric environment for certain drugs prone to degradation at low pH
- Protecting the stomach lining from certain types of drugs
- Improving bioavailability of drugs that are predominantly absorbed in the proximal small intestine by designing the formulation to break apart rapidly in the duodenum only after gastric emptying
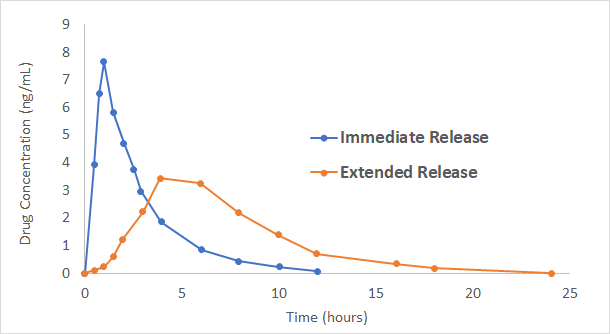
Formulation Technologies at Vici
- Polymer matrix tablets that are simple to manufacture and allow extended release
- Matrix tablets coated with a barrier membrane to closely approximate zero-order-release, often considered the gold standard for extended release
- Coated multi-particulate capsule products that allow for a variety of drug release and pharmacokinetic profiles in addition to the added benefit of sprinkle indication for pediatric applications and for the benefit of patients experiencing dysphagia.
- Combining immediate and extended-release concepts in the same formulation to achieve quick and lasting effect. This may be achieved using tablets or multi-particulate bead filled capsules.
- Osmotic tablets for very precise, gastric-environment independent drug release. This technology allows for achieving true zero-order release and even ascending profiles where the rate of drug release increases over time.
Formulations targeted towards pediatric patients or patients experiencing swallowing difficulties
- Liquid solutions or suspensions are formulated for improving swallowability
- Liquid formulations may be designed to be delivered through a G-tube or NG-tube. Vici has experience designing such formulations and performing needed studies to secure relevant label claims.
- Vici also has experience developing unit-dose packaging for liquid formulations for patient and healthcare provider convenience.
- Chewable and ODT formulations may be developed for when liquid formulations are not preferable or feasible.
- Powders or granules for reconstitution may also be formulated when drug stability concerns exist for certain molecules.

Facility and Equipment
Vici has a variety of equipment suited for developing oral dose formulations. Vici will acquire any R&D equipment needed for formulation development if there is a specific client need we cannot currently meet.
Phone
Location
6655 Amberton Drive,
Unit O, Elkridge, MD 21075